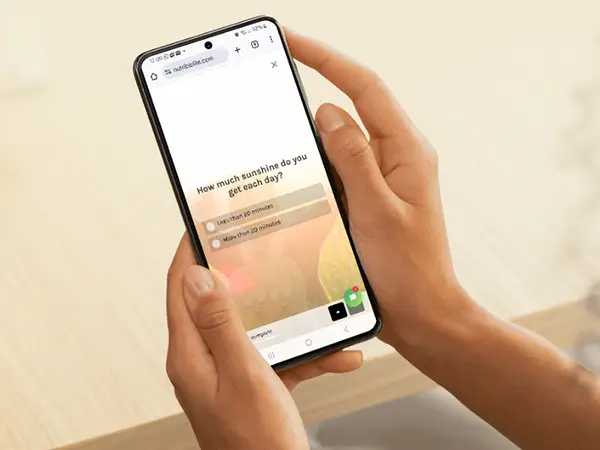
Take the Nutribiolite Quiz
Answer a few simple questions to discover the right supplements, tailored to your body, lifestyle, and health goals.

Join Our Health Club
Become a member of the Nutribiolite Health Club and get access to health insights, and special offers, starting with 5% off your first order.
Thank you!
Welcome to our community!
As a valued member, you now have access to exclusive offers and expert advice to enhance your health and wellbeing. Check your email for your discount code—if it’s not in your inbox, please look in your Spam folder. To ensure you don’t miss out on any future offers and important health updates, mark our emails as safe.
We’re excited to support you on your journey to better health!
Free shipping for selected EU countries
We process and ship all orders within 24–48 hours. Enjoy free shipping on orders over €65. For more details, please see our Shipping & Returns policy.